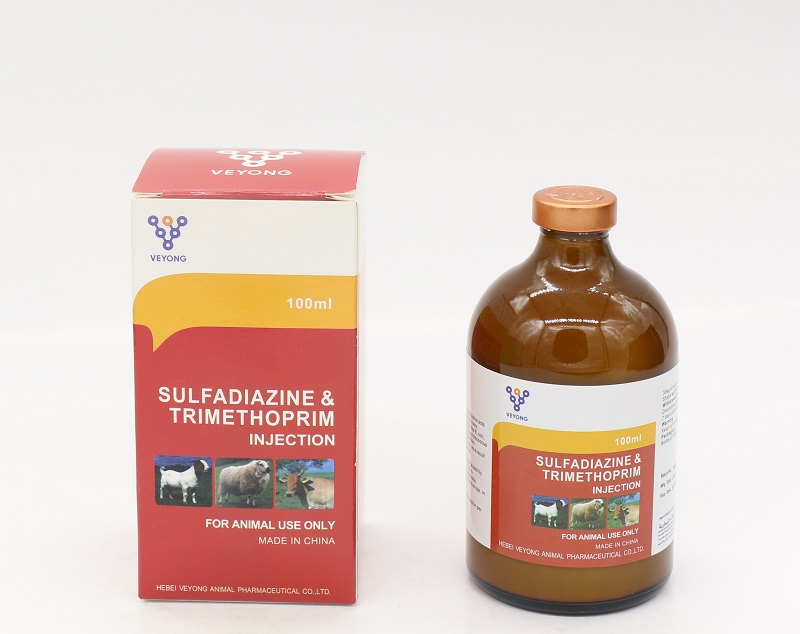
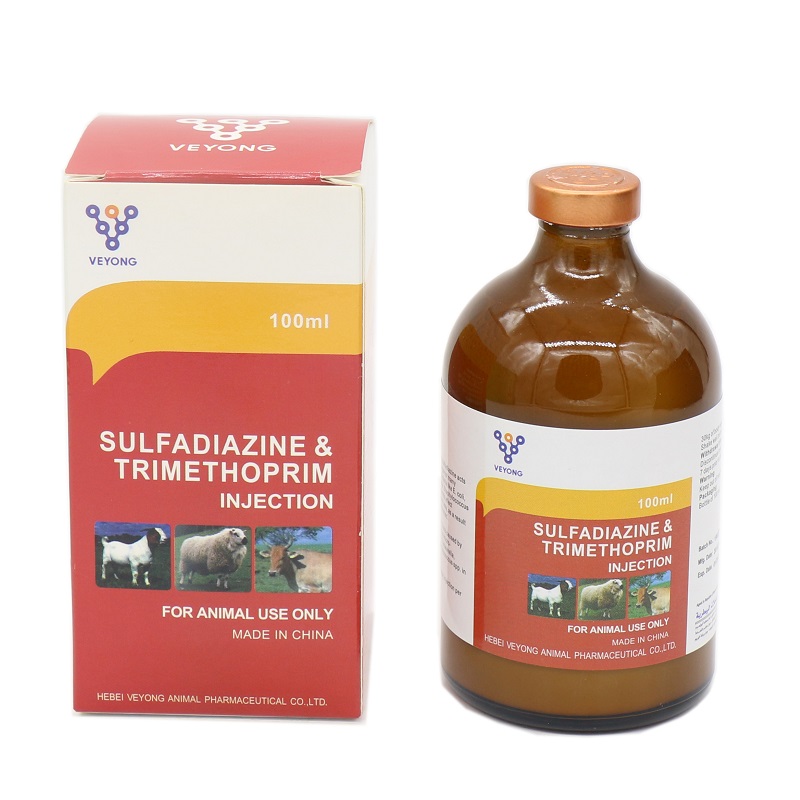
20% Sulfadiazine + 4% Trimethorim injection
Mea hana ikaika
Sulfadiazine 20.00% w / v.
Trimetopprim 4.00% w / v
Hana pharmacological
ʻO Sulfadiazine kahi lāʻau lapaʻau maikaʻi maikaʻi loa no ka hoʻohanaʻana i ka'ōnaehana'ōnaehana ʻO kāna hana hana o ka hana no ka mea he mea kūpono ia e like me ka p-aminobenzoic i ka synthesia e hana ai ma keʻano he banteria.
Indlcation
Hōʻikeʻia kēia hopena i loko o ka mālamaʻana i nā maʻi o nā'ōnaehana'ōnaehana ma muli o nā organisms e pili ana i ka trimetopprim: sulfoadiazine. ʻO ka hiʻohiʻona o ka hana e pili ana i nā mea a me nā mea maikaʻiʻole a me nā mea maikaʻiʻole a me nā mea maikaʻiʻole. ʻO Kleberiella SPP, Titsurella Spp, Pneumococci. ʻO ka proteususa, paʻakai sppp. Staphylococci, Strctococci, vibrio.
Dosage a me ka hoʻokele
E ka hopena subcutaneous wale nō.
ʻO nā holoholona:ʻO ka helu Dose i'ōleloʻia he 15 mg o nā mea hana ikaika no ka momona o ke kino o ke kino (1 ml i ka paona
Horses
ʻO nā'īlio a me nā'īlio:ʻO ka helu Dose i'ōleloʻia he 30mg o nā mea hana ikaika no kēlā me kēia kino kino (1 ML no ke kino kino).
Nā hoʻohālikelike
ʻAʻole e hāʻawiʻia i ka pahu i nā ala'ē aʻe ma mua o nā mea i hāʻawiʻia.
ʻAʻole e lawelaweʻia e hoʻopiliʻia, intra - arterial a iʻole intrathecally.
Mai lawelawe i nā holoholona me nā holoholona me kaʻike sulphomimivies,ʻoi aku ka nui o ka paleʻana i ka paleʻana o ka parenfymal
Nā Waʻa'ōlelo '
1 No ka hoʻokeleʻana o ka huahana Intravenous i keʻano o ke kino a me keʻano lohi ma mua o ka manawa kūpono.
2 I ka hōʻailona mua o kaʻaeʻana i ka inlerance i ka injection e hoʻopiliʻia a me ka mālamaʻana i ka mālamaʻana.
Pono e lawa ka wai inu e pono ai i ka wā o ka hopena o ka hana o ka huahana.
Ka haʻaleleʻana i ka manawa
Holoholona:ʻO kaʻiʻo - 12 mau lā
Milla - 4 mau lā.
Huanui
Pale aku mai ka lā pololei a mālama i lalo o 30 ℃.
He mamo na ia ka waiwai o Hebei Veyong Parman ʻO ia kahi mea nui GMP-hōʻoia i hōʻoiaʻia, me R & D, hana a me ke kūʻai akuʻana i nā mea kanu a me nā mea kanu maikaʻi. Me ke kikowaena kikowaena kikowaena, ua hoʻokumuʻo Veyong i kahi'ōnaehana R & D no ka lāʻau lapaʻau hou, aʻo ia ka mea hana pilikino iʻikeʻia, aia he 65ʻenehanaʻenehana. He mau hanaʻelua o Veyong: shijiazhuang a me ke kohoʻana o ka saijiazulang, a me nā huahana hana he 11,706 ʻO nā Pestitides a me nā mea hoʻowahāwahā, nā ents. Hāʻawiʻo Veyong i nā apis
Hoʻokomoʻiaʻo Veyong i ka mea nui i ka hoʻokeleʻana o EHS (kaʻenehana a me ke olakino a me ka palekana) a loaʻa i nā palapala ISO140018001800180018001 Ua kākau inoaʻo Veyong i kaʻoihanaʻoihanaʻoihanaʻoihanaʻoihana i nāʻoihanaʻo Hebei a hiki ke hōʻoia i ka hopena mau o nā huahana.

Ua hoʻokumuʻo Veyong i ka'ōnaehana hoʻokele kūpono piha, loaʻa i ka palapala hōʻoia ISO9001,ʻo China GPmp Loaʻa iā Veyong i ka hui o ka Region, kūʻai a me ka lawelaweʻoihana, ua loaʻa i kā mākouʻoihana i nā mea kūʻai aku maikaʻi loa a me ke kūʻai aku. Ua hana maikaʻiʻo Veyong i ka hanaʻana me nā mea honua he nui iʻikeʻia e nā holoholona hihiu,ʻAmelika Hema,ʻoʻAmelika a me nā'āina waena.




.png)
.png)
.png)
.png)